- Drug Topics November/December 2023
- Volume 167
- Issue 10
Roflumilast Foam 0.3% Secures Major FDA Approval to Treat Seborrheic Dermatitis
The foam formulation and once-daily application of roflumilast foam 0.3%, the first FDA-approved drug for seborrheic dermatitis with a new mechanism of action in over 2 decades, makes it a favorable alternative to existing treatments.
Arcutis Biotherapeutics announced today the FDA approval of roflumilast foam 0.3% (ZORYVE) for the treatment of seborrheic dermatitis in patients aged 9 years and older.1 Most notably, roflumilast foam 0.3% is a once-daily steroid-free foam and the first FDA-approved drug for seborrheic dermatitis with a new mechanism of action in over 2 decades.
“Seborrheic dermatitis is one of those issues that seems trivial and can be dismissed, but causes a great deal of distress and discomfort to patients. The itch, the redness, and the flaking cause significant impact on quality of life. Making things more difficult, many of our existing treatments are formulated as creams, ointments, or oils. While those can be ideal in some scenarios, for many patients these are extremely unpleasant on the scalp,” Peter Lio, MD, clinical assistant professor of dermatology and pediatrics at Northwestern University Feinberg School of Medicine, told Dermatology Times. "To have a new, cosmetically elegant foam formulation and a once daily application is very exciting. Moreover, a non-steroidal medication like roflumilast seems ideal for this role, with a very favorable safety profile and the ability to use it on the face for longer periods of time safely.”
Roflumilast foam provides rapid disease clearance and significant reduction in itch. In the roflumilast foam-treated arm of the pivotal phase 3 STRATUM study (
“Most patients have itch, scaling, and redness that is very unpleasant and can be tough to address given the hair-bearing nature of the scalp. Many patients express frustration with oil formulations--that it leaves the hair greasy and flat, that it stains pillowcases, and that it is really unpleasant to use. Many patients also desire non-steroidal treatment options, especially for those that also have seborrheic dermatitis involvement on the face. While not necessarily a replacement, a non-steroidal option can be used intermittently to give breaks from steroids in some patients as well,” said Lio.
In exclusive interviews with Dermatology Times, Shawn Kwatra, MD, and Christopher Bunick, MD, PhD, discussed the significance of the historical approval and the unique mechanism of action of roflumilast foam as a phosphodiesterase-4 (PDE4) inhibitor.
Celebrating a Historic Moment
Shawn Kwatra, MD, associate professor of dermatology at Johns Hopkins University School of Medicine and director of the Johns Hopkins Itch Center, in Baltimore, Maryland, emphasized the incredible significance of roflumilast foam’s approval for seborrheic dermatitis, a common skin condition with no specific FDA-approved therapy.
“We have to also applaud Arcutis as a company for being able to take a chance going in on a new indication and being able to help develop the field, being able to validate instruments, and taking that chance. I think it's very important for dermatology that we advance as a field with new therapeutics for our orphan conditions. I've seen this happen in prurigo nodularis. It's very exciting to have it happen in seborrheic dermatitis because oftentimes, when there's a major investment made in understanding a disease and clinical trials, then we end up learning a whole lot more about what's really going on. So, this is a very historic day for us that we have a new therapeutic,” said Kwatra.
Diving Into the Mechanisms of Action
Christopher Bunick, MD, PhD, associate professor of dermatology and physician-scientist at the Yale School of Medicine in New Haven, Connecticut, discussed in-depth the mechanism of action of roflumilast foam and its ability to boost cAMP, and therefore create an anti-inflammatory effect.
“That's why seborrheic dermatitis patients are going to see a very good reduction in the redness of the skin lesions, and this will also reduce the scaling effect from the inflammation itself. What makes roflumilast’s mechanism of action really stand out is that at a molecular level, roflumilast is anchored into the binding site of PDE4 by 3 major contacts. Those 3 major contacts help give it that high binding affinity that translates, we think clinically, into the potency that we see with roflumilast,” said Bunick.
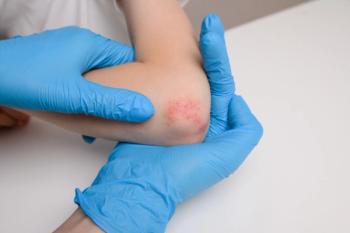
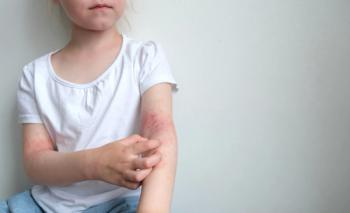
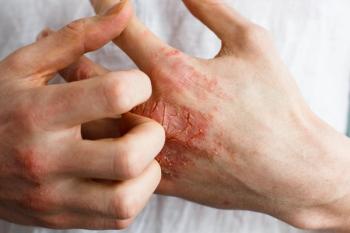
Seborrheic dermatitis causes red patches covered with large, greasy, flaking yellow-gray scales, and persistent itch. In patients with skin of color, inflamed areas may not appear red, but instead can appear pink, slightly purple, or lighter in color than the surrounding skin. Seborrheic dermatitis occurs in areas of the body with sebaceous glands, including the scalp, face (most prominently on the nose, eyebrows, ears, and eyelids), upper chest, and back. Due to the nature of hair around these areas, topical creams, gels, and ointments are difficult to apply.
According to the announcement, Arcutis intends to make roflumilast foam widely available via key wholesaler and dermatology pharmacy channels as a new treatment option by the end of January 2024. The
The approval of roflumilast foam is based on positive results from the phase 2 Trial 203 and phase 3 STRAUM trial. Both trials were parallel-group, double-blind, vehicle-controlled studies evaluating the safety and efficacy of roflumilast foam 0.3% in seborrheic dermatitis. Overall, both studies enrolled 683 adults and adolescents aged 9 years and older.
The STRATUM study met its primary endpoint, with nearly 80% of patients treated with roflumilast foam achieving Investigator Global Assessment (IGA) Success rate at week 8 (79.5% roflumilast foam vs 58.0% vehicle; P<0.0001). In Trial 203, 73% of patients treated with roflumilast foam achieved IGA Success (73.1% roflumilast foam vs 40.8% vehicle; P<0.0001.) IGA Success was defined as an IGA score of “Clear” (0) or “Almost Clear” (1), plus a 2-grade IGA score improvement from baseline at week 8.
Data from the STRATUM study also demonstrated statistically significant improvement over vehicle on all secondary endpoints, including itch, scaling, and erythema. More than 60% of patients achieved a ≥4-point reduction in itch at week 8 as measured by Worst Itch-Numerical Rating Score (62.8% roflumilast foam vs 40.6% vehicle; P=0.0001), and significant improvements in itch were also reported at week 2 and week 4. Patients treated with roflumilast foam reported a 28% improvement in itch from baseline in 48 hours (compared to 13% on vehicle nominal P=0.0024).
Roflumilast foam was well-tolerated with a favorable safety and tolerability profile during up to 52 weeks of treatment. Incidence of Treatment Emergent Adverse Events (TEAEs) was low and similar between active treatment and vehicle, with most TEAEs assessed as mild to moderate severity. There were no treatment-related Serious Adverse Events (SAEs). Overall, the most common adverse reactions occurring in ≥1% of patients in the combined phase 2 and phase 3 study populations were nasopharyngitis (1.5%), nausea (1.3%), and headache (1.1%).
“Approximately 10 million people in the United States have seborrheic dermatitis, but until today, there have been limited treatment options. We are thrilled with this FDA approval and are excited to bring to market a new, highly effective steroid-free topical formulation that can be used anywhere on the body. Our commercial team is ready and poised to launch ZORYVE foam very soon, and we are committed to ensuring affordable access to ZORYVE foam to those who may benefit from this novel treatment,” said Frank Watanabe, the president and CEO of Arcutis, in the news release.1
Reference
1. FDA approves Arcutis’ ZORYVE (roflumilast) topical foam, 0.3% for the treatment of seborrheic dermatitis in individuals aged 9 years and older. News release. Arcutis Biotherapeutics. December 15, 2023. Accessed December 15, 2023. https://www.arcutis.com/fda-approves-arcutis-zoryve-roflumilast-topical-foam-0-3-for-the-treatment-of-seborrheic-dermatitis-in-individuals-aged-9-years-and-older/
Articles in this issue
almost 2 years ago
PA Programs Draw Pharmacy’s Best and Brightestabout 2 years ago
Expert Interview: Protein-Based Vaccines for COVID-19over 2 years ago
In the Opioid Epidemic, Treatment Challenges Still PersistNewsletter
Pharmacy practice is always changing. Stay ahead of the curve with the Drug Topics newsletter and get the latest drug information, industry trends, and patient care tips.